Partnership Opportunities
Following an exciting year of multiple first-in-class approvals, more than 2000 cell and gene therapies currently in development, investment is flooding into gene therapy pipelines. Are you ready to capitalise on the booming gene therapy landscape?
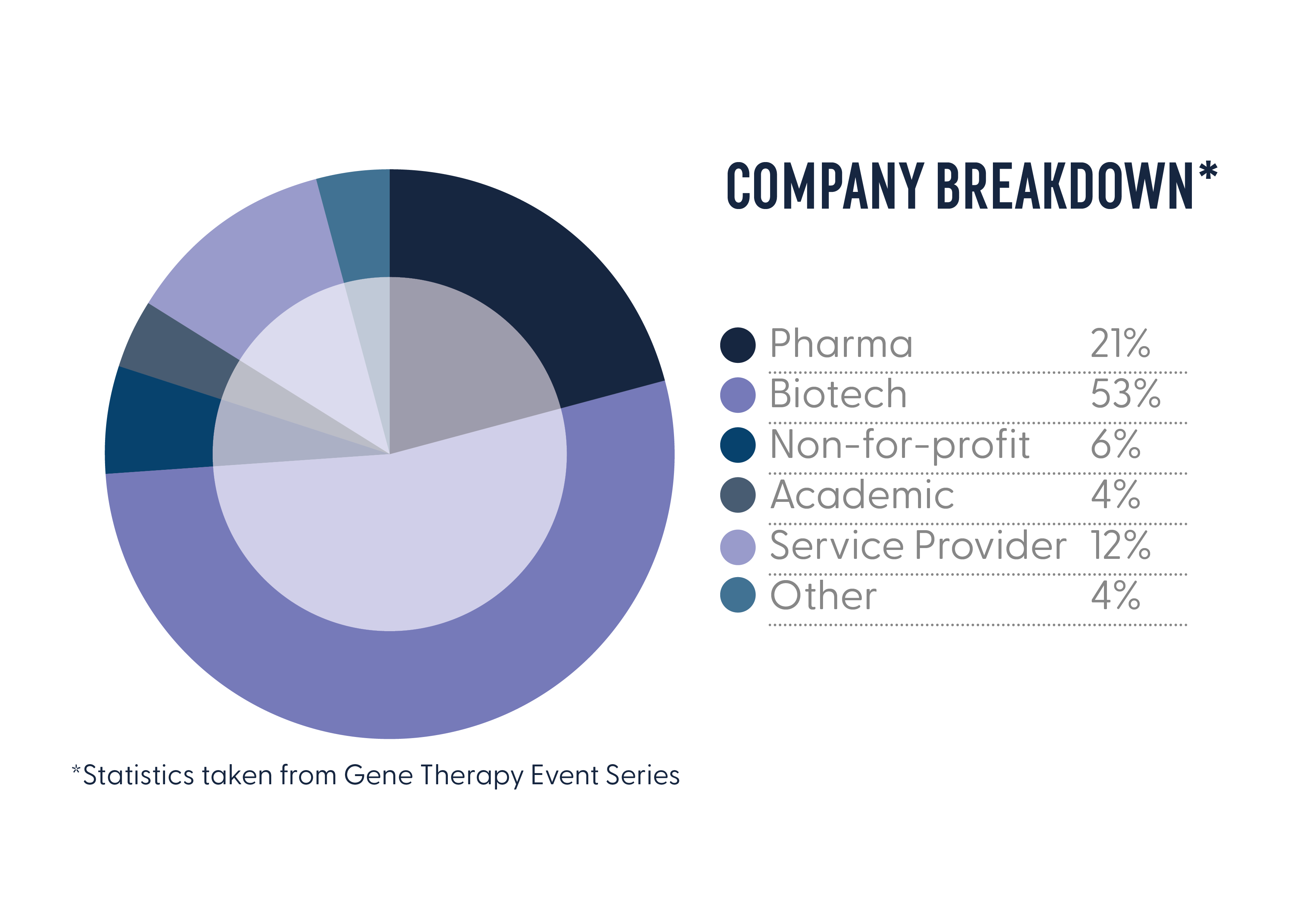
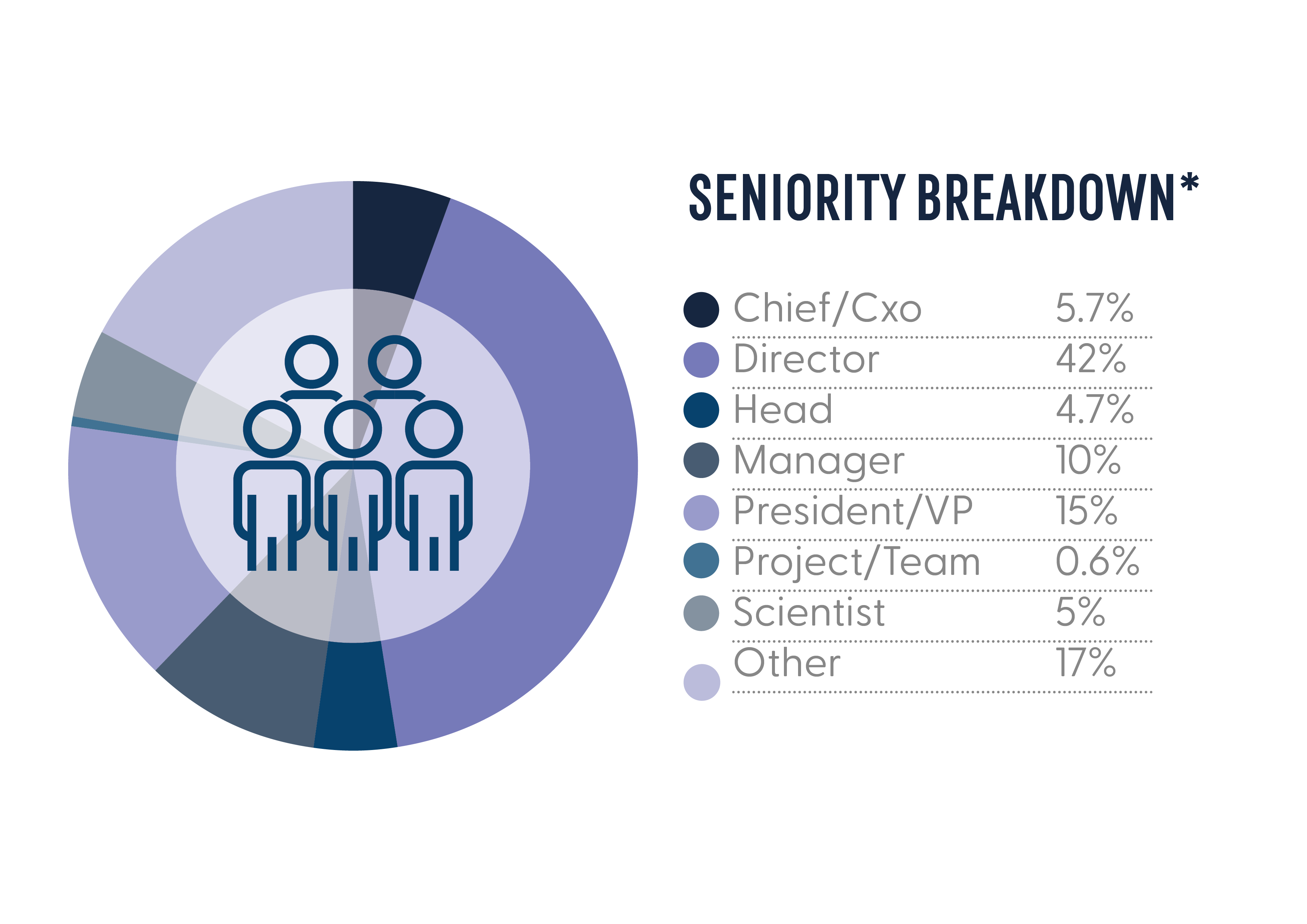
In January 2024, the Gene Therapy Regulatory Affairs forum united 80+ of decision-makers across large-pharma, leading and emerging biotech’s with expertise in clinical, non-clinical and CMC regulatory affairs. As the only event of its kind, this was a unique and valuable opportunity to showcase your work to an exclusive audience of regulatory expertise in gene therapy.
There are a limited number of partnerships available, including speaking positions across our 2025 agenda. If you provide regulatory consultancy services or support with clinical trial design (CRO) that would benefit our niche audience, now is the time to secure new business and form lasting connections with the companies seeking strategic and detailed regulatory solutions to progress their products to market.
Work with us to create a bespoke, tailor-made partnership to achieve your business and strategic goals, and help you meet the key stakeholders you want to work with to put the spotlight on your service.
Experts Need Your Help With:
Expert regulatory consultants
in clinical, non-clinical and CMC, to support functions ranging from technical writing and regulatory strategy, to IND, GMO & BLA applications and more
Clinical trial design
CROs to perfect trial strategy for regulatory success, for project management and program development
Why Partner?
Take the mic on the main agenda to build confidence with the audience and showcase your specific expertise, distinguishing your unique value proposition from your competitors
Build brand awareness and visibility through our pre-conference and in-event advertising
Win new business by meeting key stakeholders organising partnerships during our speed-networking sessions and organised 1-2-1 conversations with the companies you want to work with
Cement your company as the leading provider for your service in gene therapy research and regulation
Benefit from our exclusive market insights about what your customers are looking for
Who Could You Meet in 2025?